
“These data reaffirm the importance of testing for PD-L1 expression in non–small cell lung cancer in order to identify those patients who are most likely to benefit from treatment with Keytruda.” 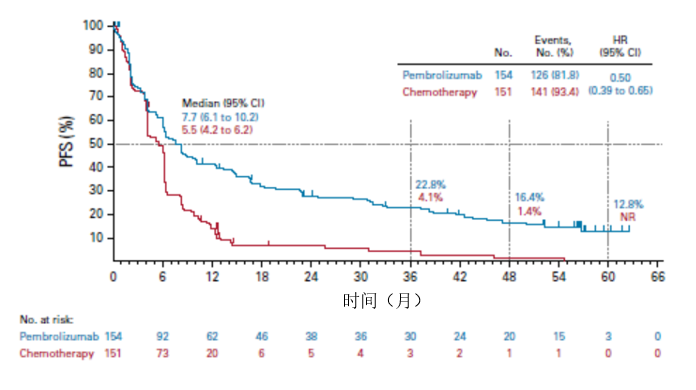
Herbst, MD, PhD, professor of medicine and chief of medical oncology, Yale Cancer Center and Smilow Cancer Hospital at Yale New Haven, said in a statement. “With this new indication, Keytruda can now be a first treatment option instead of chemotherapy for patients with metastatic nonsmall cell lung cancer whose tumors express high levels of PD-L1,” Roy S. The action converts the accelerated approval pembrolizumab previously received in this setting to a full approval. 1,2Īlong with the frontline approval, the FDA also authorized an update to pembrolizumab’s label to include data from the KEYNOTE-010 trial, 3which examined the PD-1 inhibitor in the second-line setting and beyond for patients with NSCLC and PD-L1 expression levels of ≥1% who have progressed on platinum-based chemotherapy and EGFR- or ALK-targeted therapy for individuals harboring those aberrations. The approval is based on data from the phase III KEYNOTE-024 trial, in which single-agent pembrolizumab reduced the risk of death by 40% and improved progression-free survival (PFS) by 4.3 months compared with doublet chemotherapy for untreated patients with advanced NSCLC with PD-L1 expression on ≥50% of cells. Pembrolizumab (Keytruda) has received FDA approval for the frontline treatment of patients with metastatic nonsmall cell lung cancer (NSCLC) whose tumors have ≥50% PD-L1 expression based on an FDA-approved test and who do not harbor EGFR or ALK aberrations.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
